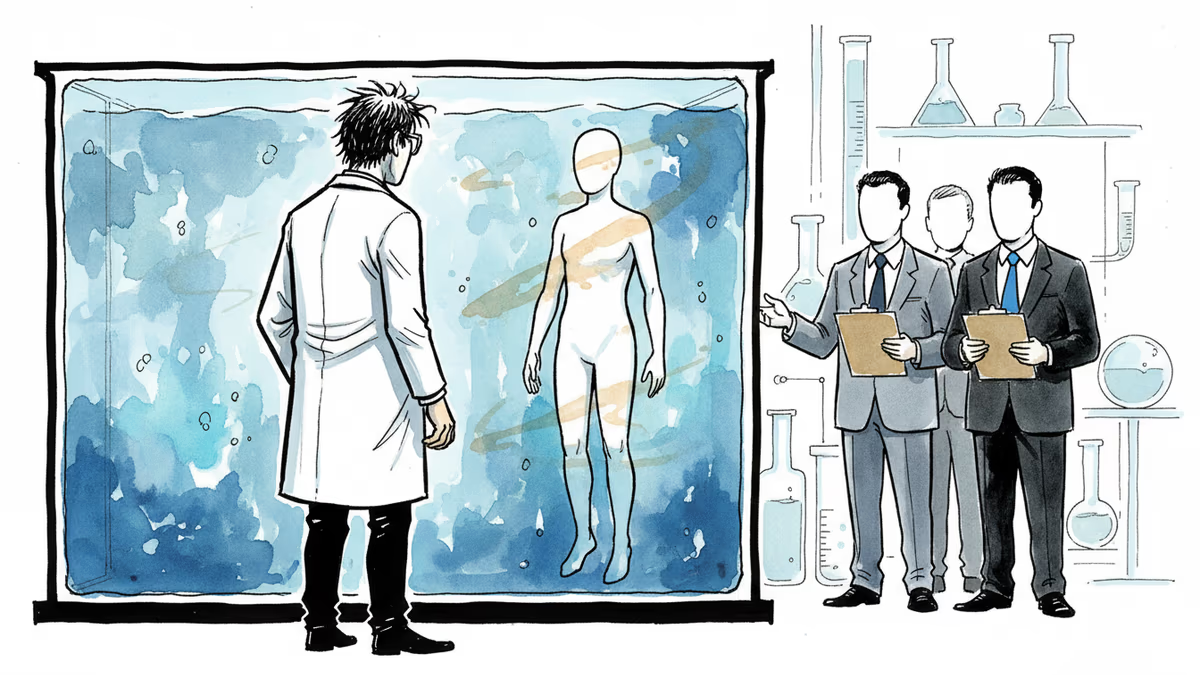
Someone Raised Money to Build Brainless Human Clones
California startup R3 Bio quietly pitched investors on 'brainless human clones' as backup bodies—beyond its public story of nonsentient organ sacks for drug testing. A deep dive into biotech's most ethically charged frontier.
Somebody heard the pitch. Somebody wrote the check.
That's the detail that makes this story different from a science fiction premise.
R3 Bio, a California-based startup, spent years operating in near-total secrecy. Last week, it broke cover—but only partially. The official reveal was striking enough on its own: the company said it had raised funding to develop nonsentient monkey "organ sacks," essentially brainless primate bodies grown as an alternative to conventional animal testing in drug development.
Then MIT Technology Review started asking questions.
The Pitch Behind the Pitch
What founder John Schloendorn presented to investors went considerably further than monkeys. According to MIT Technology Review's reporting, Schloendorn pitched a vision of "brainless human clones" grown as backup bodies—biological spare-parts depots, genetically matched to their owners, available for organ harvesting without immune rejection.
R3 Bio did not want this reported. The company declined to engage with the publication's inquiries.
The significance here isn't just the concept itself—it's the context. This wasn't a thought experiment floated at a conference. It was a funding pitch. Which means investors evaluated it, and at least some decided it was worth backing.
The line between speculative biology and venture-backed reality just moved.
Meanwhile, a Uterus Survived Outside the Body
In a separate but thematically resonant development, reproductive health researchers have kept a donated human uterus alive outside the body for one full day using a device they've named "Mother." The organ was connected to the machine's artificial circulatory system ten months ago, with modified human blood pumped through it continuously.
The team describes this as a proof-of-concept—the first time a human womb has been maintained ex vivo. Future iterations, they suggest, could illuminate the mechanics of pregnancy, and potentially, someday, support fetal development entirely outside a human body.
Taken together, these two stories aren't coincidental neighbors in a news cycle. They're data points on the same trajectory: the technical decoupling of human biological function from the human body as a whole.
Three Ways to Read This
For patients and the medical community, the underlying problem R3 Bio is trying to solve is genuinely urgent. Over 100,000 people in the US alone are on organ transplant waiting lists at any given time. Thousands die each year before a compatible donor becomes available. A supply of immunologically matched organs—however they're grown—would represent a meaningful clinical advance. The logic is hard to dismiss.
For bioethicists, the architecture of the argument is precisely what's troubling. The entire framework rests on the premise that absence of a brain equals absence of moral status. But consciousness remains one of the least understood phenomena in science. What happens if a "brainless" clone develops unexpected neural activity? What legal category does it occupy? The answers don't exist yet—and the technology isn't waiting for them.
For investors and the longevity industry, this sits at the intersection of several enormous markets: organ transplantation, drug testing platforms, and the rapidly expanding field of life extension for high-net-worth individuals. The fact that R3 Bio secured funding while operating in secrecy suggests that capital is already moving into this space, away from public scrutiny and regulatory attention.
The Regulatory Vacuum
Human cloning is prohibited under law in many jurisdictions—but those laws were written with a different technology in mind. Whether a deliberately brainless human-derived organism constitutes a "clone" in the legal sense is not a settled question. The gap between what the science can attempt and what the law clearly governs is widening.
The FDA regulates cell and tissue products, but the framework for evaluating something like a nonsentient human-derived organ system doesn't cleanly exist. Europe's regulatory environment is stricter on bioethical grounds, but enforcement depends on where the research is conducted—and R3 Bio is in California, where the rules are somewhat more permissive for early-stage research.
The startup's instinct to stay quiet makes a certain strategic sense: move fast, establish facts on the ground, and let regulators catch up.
Authors
Related Articles

R3 Bio wants to grow human bodies without brains as biological backups. The science is speculative. The ethics are explosive. And the legal framework doesn't exist yet.

Colossal Biosciences raised hundreds of millions to resurrect woolly mammoths and dire wolves. What they're actually doing is more complicated—and more interesting.

Around 5,000–6,000 people worldwide have signed up to have their bodies or brains cryonically preserved after death. Scientists say revival is vanishingly unlikely. So what exactly are they waiting for?

Max Hodak's Science Corp raises $230M for rice-grain sized chip that restores vision to blind patients. Could beat Neuralink as first BCI company to commercialize
Thoughts
Share your thoughts on this article
Sign in to join the conversation